No products in the cart.
LIVE - Rebellion Reads Posts
Tattoo Ink Regulations: REACH & The REBEL Shield
Tattoo Ink Regulations: Banned in Berlin, Legal in Vegas
Tattoo Ink Regulations have a fundamental customer protection purpose. In tattooing, the client pays for two things at once: the artist’s skill and a product that stays in their skin for life. Therefore, Tattoo Ink Regulations are not abstract policy—they directly affect what the tattoo artist implants into another human body.
The Illusion of Oversight in Tattoo Ink Regulations
Most clients assume tattoo ink goes through the same testing as medicine or food. Thus, their health concerns usually stop at sterilization and bloodborne pathogens. As a result, they rarely question the ink itself.
In the United States, regulators classify tattoo inks as cosmetics. However, cosmetics sit on the surface of the skin and can be removed, like makeup or body lotions. Tattoo ink, by contrast, is an intradermal application where a foreign particle is injected into the dermis with the intention of remaining there permanently. Consequently, the regulatory categorization does not match the biological reality.
That gap creates a misunderstanding from the start.


Product vs. Service: The disconnect
When someone buys food or a drink, they can read the ingredient list, compare brands, and make an informed decision about purchasing it or not. Moreover, they physically hold the product in their hands before consuming it.
Tattooing works differently. The client purchases a service. Meanwhile, the ink bottle never reaches their hands. They do not have the chance to read the ingredient lists or check compliance documents. Instead, they evaluate the artist’s portfolio, trust the reputation, and agree on a price.
Therefore, tattoo collectors pay for a permanent implanted product they never personally examine.
This explains why many people assume, “If it’s on the shelf, it must be cleared.” However, Tattoo Ink Regulations in the U.S. do not function like food labeling laws.


Global Tattoo Ink Regulations: EU vs. US
The European Union regulates tattoo inks under REACH. Under this framework, authorities restrict substances when credible suspicion of risk exists. In other words, the EU applies a precautionary principle.
By contrast, the U.S. model usually requires demonstrated harm before regulators restrict a substance. Therefore, the American system reacts to evidence rather than preventing potential exposure in advance.
This philosophical difference creates real-world consequences. For example, an ink legal in Las Vegas may face restriction or ban in Berlin. The formula may be identical; however, the regulatory threshold differs.
Artists who source internationally must understand that Tattoo Ink Regulations depend on jurisdiction, not just chemistry.


Pigments: From Plastic Bag to Tattoo Ink
Many believe that tattoo pigments are designed specifically for human injection. In reality, many base pigments were originally formulated for industrial applications, such as coatings (car and wall paints) or commercial printing (printer ink). A black plastic bag, a phone case, and a bottle of tattoo ink may share the same pigment origin; the critical difference lies in the purification process. Achieving pharmaceutical-grade purity is a costly endeavor, and it is precisely where manufacturers cut corners to increase profitability.
Today, responsible ink production focuses on selection, purification, and impurity control. Manufacturers must monitor heavy metals, aromatic amines, and other trace contaminants. Therefore, modern formulation discipline matters more than ever. Current research shows associations, mechanisms, and increased risks among other factors.
Current Tattoo Ink Regulations increasingly scrutinize impurity profiles rather than just color identity. As a result, formulation integrity becomes a measurable standard, not a marketing claim.
Tattoo Ink and the Body: Biological Perspective
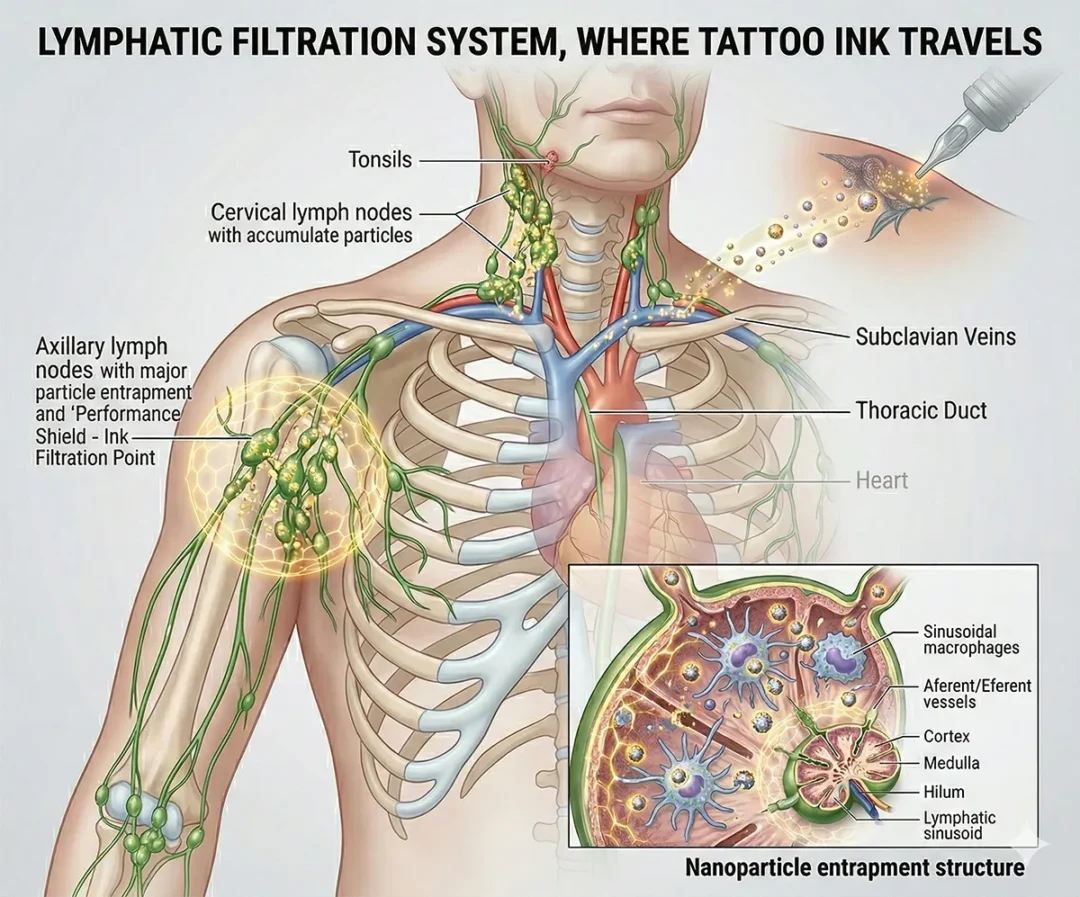
Tattooing is an intradermal application. It introduces foreign particles into living tissue. Immediately, the immune system responds. Macrophages engulf pigment particles and attempt to contain them. Larger particles remain in the dermis. Smaller particles (approximately < 50 nm), however, migrate through lymphatic pathways.
Researchers have documented nanoparticle migration and pigment accumulation in regional lymph nodes. The lymph nodes of tattooed individuals become “tinted” or stained with the same color as the tattoo itself, and pigment particles migrate rapidly. Studies show that pigment particles enter nearby lymph nodes within minutes of the tattooing process. Furthermore, research demonstrates long-term immune interaction in those nodes. While most pigment remains in the skin, a measurable portion is transported from the dermis to the lymph nodes and reaches other organs, including the liver, spleen, and lungs, via the bloodstream.
Therefore, ink formulation has biological implications beyond visible color. This is why honest manufacturing matters.
Responsibility in a Service-Based Industry
Tattooing operates as a service-based business model in which a product is permanently implanted into the body. Because clients cannot inspect the ink formulation themselves, they ultimately bear the consequences of whatever purchasing decisions the tattoo artist makes—whether those decisions are informed, disciplined, or purely price-driven.
Responsibility operates on three levels:
- Government establishes the regulatory structure.
- Manufacturers formulate and document product safety.
- Artists decide what they purchase and implant into their clients.
Consequently, ink selection is not a minor supply choice; it directly affects client exposure and long-term risk.
The REBEL Performance Shield
In the U.S. market, purchasing decisions follow a predictable order: price first, performance second, client protection third. Ignoring that hierarchy would be unrealistic.
REBEL integrates them all.
- Performance: Artists need reliable, one-pass saturation. Efficient application reduces overworking and protects profitability. Time equals revenue.
- Value: Competitive pricing matters. However, manufacturers should not cut corners in raw material purification or impurity control to reach that price point.
- Built-In Client Protection: Formulations aligned with strict impurity thresholds—such as EU REACH standards—add an additional layer of discipline. Even when U.S. law does not require those limits, responsible manufacturers can adopt them voluntarily.
- Future Proof Formulation: Formulated in alignment with advanced international impurity thresholds, independent of current U.S. federal requirements.
The competitive edge is straightforward: not all inks meet the same internal standards. Artists who understand Tattoo Ink Regulations can choose accordingly.


Tattoo Ink Regulations: The Bottom Line
Tattoo Ink Regulations continue to evolve. On one hand, MoCRA increases accountability in the United States. On the other hand, REACH enforces stricter precautionary controls in Europe. Meanwhile, scientific research confirms systemic pigment migration in the human body.
Tattooing remains a service. Clients pay for expertise and for a permanent product that never passes through their hands and bear the potential health consequences.
Therefore, the ink in the bottle matters.
Spread the Knowledge
If this article gave you clarity, share it with fellow tattoo artists and apprentices. The more we understand how our tools are built, the higher we can set the standards of tattooing.
Make Your Voice by Leaving a Comment
Got questions or insights about today’s topic? Drop them in the comments — your voice helps push the craft forward.